Under the Examination Guidelines of the Japan Patent Office (JPO), an application for patent term extension based on a drug approval will be rejected when the drug product that is the subject of the drug approval is not different from that of the prior approval in elements (features) recited in the patented claim. The English Examination Guidelines are available from the website of the JPO (please see “Part VI Patent Term Extension”).
However, the grand panel of the Intellectual Property (IP) High Court negated the current practice of the JPO. The summary of the grand panel decisions will be shown below. The English translation of the judgment will soon be available from the website of the IP High Court.
The JPO Commissioner has appealed to the Supreme Court. Accordingly, the grand panel decisions have not yet become final and binding. However, there is a possibility of an increase in the cases where a patent term is allowed to be extended. Instead, the extent of a patent right during an extended patent term may be narrowed.
Case Numbers:
(1) 2013(Gyo-Ke)10195 (appealed from Fufuku2011-8105, JP 3398382 B (‘382 Patent))
(2) 2013(Gyo-Ke)10196 (appealed from Fufuku2011-8106, ‘382 Patent)
(3) 2013(Gyo-Ke)10197 (appealed from Fufuku2011-8107, JP 3957765 B (‘765 Patent))
(4) 2013(Gyo-Ke)10198 (appealed from Fufuku2011-8108, ‘765 Patent)
Parties:
(Plaintiff) GENENTECH, INC.
(Defendant) JPO Commissioner
1. Factual background:
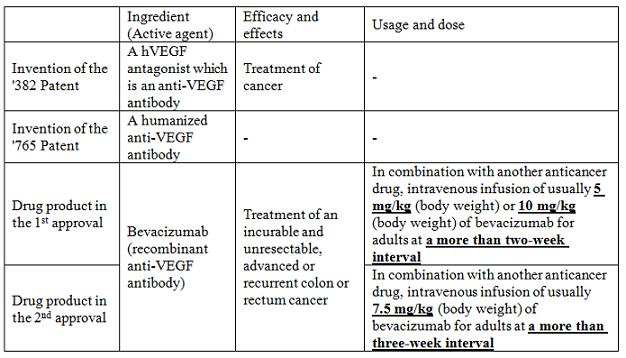
The Plaintiff is a Patentee of the ‘382 Patent and ‘765 Patent, and filed applications to request the extension of their patent terms for 5 years for the reason that a drug approval under the Pharmaceutical Affairs Law was necessary for the working of the patented invention. The inventions of the ‘382 Patent and ‘765 Patent are as follows:
[Claim 1 of the’382 Patent]
A composition for treatment of cancer comprising a therapeutic amount of a hVEGF antagonist which is an anti-VEGF antibody.
[Claim 1 of the ‘765 Patent]
A humanized anti-VEGF antibody which binds human vascular endothelial growth factor (VEGF) with a Kd value of —–, having a heavy chain variable domain comprising —–, and a light chain variable domain comprising —–.
There was another drug approval (the 1st approval) prior to the drug approval which was basis for the patent term extension (the 2nd approval). Drug products in these approvals are different in only “usage and dose,” which are not recited in the patented claim. The following table summarizes the patented inventions and the drug products in the 1st and 2nd approvals.
2. Reason of the Appeal Decision by the Appeal Board:
The applications based on the 2nd approval were rejected by the Appeal Board of the JPO because of the presence of the 1st approval. The Appeal Board stated in the Appeal Decision as follows:
According to Article 67-3(1)(i) of the Japanese Patent law, an application for patent term extension is to be rejected unless the approval which the application is based on was necessary for the working of the patented invention. The phrase “working of the patented invention” in Article 67-3(1)(i) should not be interpreted as production and distribution of a drug product that is the subject of an approval. Instead, it should be interpreted as production and distribution of the drug product which is defined by the “matters examined for the approval which correspond to elements (features) recited in the patented claim” (the Examination Guidelines).
The drug product defined by the “matters recited in the approval which correspond to elements (features) recited in the patented claim” (active agent and efficacy and effects) of the 2nd approval is the same as that of the 1st approval. Therefore, it is not considered that the 2nd approval was necessary for the working of the patented inventions, and the application should be rejected pursuant to Article 67-3(1)(i).
3. Summary of Grand Panel Decisions of the IP High Court:
The Plaintiff filed law suits against the decisions by the Appeal Board before the IP High Court. The Grand Panel rescinded the Appeal Decision. Specifically, the Court stated as follows:
(1) Error in the judgment pursuant to Article 67-3(1)(i):
In order to satisfy the “approval was necessary for the working of the patented invention” according to Article 67-3(1)(i), the following two requirements have to be fulfilled.
(A) Production and distribution of a drug product which is a subject of an approval was workable only after the approval (for example, the production and distribution of the drug product had not been workable even after the prior approval).
(B) The production and distribution of the drug product was covered by the patented claim.
With respect to a patent which is directed to the ingredients of a drug product (except for patents of manufacturing methods and product-by-process claims), the phrase “working the patented invention” should be interpreted as production and distribution of a drug product identified by “ingredient,” “quantity,” “usage,” “dose,” “efficacy” and/or “effects,” which are matters examined for the approval listed in the Pharmaceutical Affairs Law.
In the present case, the prohibition of production and distribution of the drug product whose usage and dose is “in combination with another anticancer drug, intravenous infusion of usually 7.5 mg/kg (body weight) of bevacizumab for adults at a more than three-week interval” had not been workable even after the 1st approval. It was workable only after the 2nd approval. Therefore, Requirement (A) is fulfilled. Since Requirement (B) is also fulfilled, there is no reason for rejection.
(2) Extent of effect of the patent right whose duration is extended according to Article 68-2 of the Patent Act:
In the case of a patented invention covering the ingredients of a drug product, it is appropriate to construe that a patent right whose duration is extended according to Article 68-2 covers the product which is defined by (i) the “ingredients (not limited to active agents)” and (ii) the “efficacy and effects” and “usage and dose” described in the drug approval.
4. Our comments:
The current Examination Guidelines concerning an application for patent term extension was revised just two and half years ago. However, the grand panel of the IP High Court negated the basic premise of the examination. The JPO still maintains the current Examination Guidelines. We need to focus on future developments.